Private Label Ostomy Bags: A Complete Guide to Building Your Own Brand
Table of Contents
Have you noticed that there are more and more people requiring long-term care around us? As people age, the market for ostomy bags is growing larger and there are more opportunities.
The main purpose of this guide is to clearly explain to you, who wants to produce private label ostomy bags, the important factors involved, such as the types of diaper bags available, how to customize them, and how to choose a good manufacturer.
What Are Private Label Ostomy Bags?
Private label ostomy bags, as the name suggests, refer to ostomy bag products manufactured and sold under their own name by retail companies or private label medical product manufacturers. Simply put, it means the ostomy bag seller finds a reliable ostomy bag manufacturer to produce the product, but then affixes their own brand label to the product.
In the ostomy bag industry, private label specifically refers to:
1. Medical device products sold under the name of a private label manufacturer, using a different brand than the original manufacturer. Hospitals and patients see your brand. You are responsible for resolving any issues that arise after product use, and customers acquired during the sales process are also your resources.
2. In terms of quality, the ostomy bags are the same as those from brand manufacturers, using the same production line and quality standards. However, the outer packaging and brand name are different; you ultimately decide on your own product packaging and brand name, thus building your own brand image.
3. Ostomy bags manufactured under a licensing agreement with the original manufacturer are sold under their own brand name. These private label ostomy bags are not simply rebranded products; their quality is legally guaranteed.

Why are private labels the preferred choice for entering the ostomy products market?
Entering the ostomy bag market presents a significant opportunity. If you’re considering how to quickly establish yourself in this industry with private label ostomy bags, adopting a private label model is the best choice. The following will explain why this model is suitable for you:
1. Faster market entry with lower risk.
Compared to developing medical-grade products from scratch, this requires significantly more time and investment. Choosing a private label manufacturing model helps avoid these obstacles: you can launch your own private label ostomy bags without the heavy R&D burden, ensuring a faster route to market for your private label ostomy bags.
- Bypassing numerous hurdles: You don’t need to do specialized R&D, handle complex compliance issues, or build your own medical-grade factory. Private label partners already possess these resources, certifications, and quality control systems. This means you don’t need to spend time building a factory; instead, you can quickly operate your own platform.
- Faster market entry: You don’t need a very long product development cycle. You can use existing formulas and products to customize and produce your product. You can bring your product to market in a very short time. This speed allows you to quickly seize market opportunities.
2. Achieving cost-effectiveness and strong profitability.
The private label ostomy bags model provides many companies with the right development path, which can reduce upfront investment while ensuring profits:
- Reduced Development Costs:Businesses don’t need to bear enormous R&D expenses; they can directly use mature products developed and tested by the manufacturer. The manufacturer distributes these costs across multiple customers, giving businesses a significant cost advantage.
- Increased Profit Margins: Own-brand models typically offer profit margins of around 50%, significantly higher than traditional retail models. This increased profit comes from reduced R&D and marketing costs.
3. Focus on Core Brand Advantages.
By outsourcing complex production and R&D tasks to suppliers, valuable resources can be concentrated on building the brand:
- Enhanced Brand Value: More time can be dedicated to understanding patient needs, creating a unique brand story, developing more comprehensive strategies, and building a customer base.
- Building a Unique Image: While the product itself remains consistent, it can still be customized to meet individual patient needs. Unique packaging, brand logos, and even subtle adjustments can be made. This allows you to achieve a unique effect without completely changing the product itself, highlighting its selling points and creating a deeper and stronger connection with your audience, thus shaping your own brand image.
Customizable ostomy bag types
If you decide to enter the market with private label ostomy bags, you can’t just sell a single product line. To build a successful brand, you need to offer products that meet the diverse usage requirements and medical needs of patients.
Below are some of the main types of ostomy bags that can be promoted under your own brand:
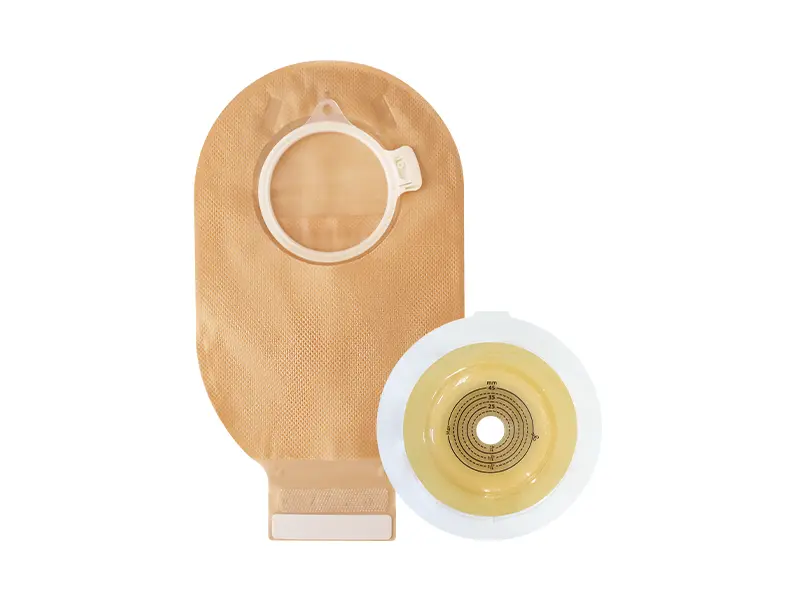
1. One-Piece System

2.Two-Piece System
What We Can Customize
In the field of medical consumables, private label ostomy bags are more than simply adding your own logo to an ostomy bag. The significance lies in producing an ostomy bag that perfectly matches your brand image and addresses the needs of patients. Zodelo can provide complete customization services in the following four key areas:
Product Specifications and Materials Engineering
The comfort and usability of an ostomy bag largely depend on its materials and components. We can precisely control the materials and components used in each product according to the patient’s needs, ensuring that every private label ostomy bags solution meets specific requirements. There are several types of film used for the bags. Some patients are particularly concerned about privacy and dislike noise, so we can choose a soft, quiet polyethylene film that moves almost silently. For those who need to monitor the stoma frequently, there are also durable, transparent models that offer clear visibility. Furthermore, the sealing components, whether clips, buckles, or sliding valves, can be customized to suit the patient’s hand dexterity, making our private label ostomy bags cater to a wider range of patient needs.
Additionally, the base plate, which directly contacts the skin, is the most critical part of the entire stoma system. We’ve invested a lot of effort in this area, visiting many patients and having them use the product, gathering feedback and continuously improving, resulting in a wide range of customization options for our private label ostomy bags. This dedication ensures that every private label ostomy bags we produce meets the highest standards of comfort and safety.
For example, with the hydrogel patch, we’ve continuously adjusted its adhesive formula. It needs to adhere firmly, last long, be sweat-proof, maintain adhesion even in cold winters, and be gentle on the skin with minimal residue when removed. We’ve struck a perfect balance.
The shape and thickness of the patch can also be varied. For patients with softer abdomens, we have smooth designs; if the stoma is slightly recessed, we have a convex design for a better fit.
We can also add thoughtful little design features, such as a more flexible ring design or a secure abdominal binder, all to help patients live like normal people.
Zodelo‘s goal is to help every patient find the perfect patch for them, one that is comfortable to use and allows for a more carefree life.
Functional Features and Accessories
The functionality of the ostomy bag directly determines the patient’s quality of life. Common problems like odor and bloating must be taken seriously. The colonostomy bag can be fitted with a waterproof activated charcoal filter, allowing for worry-free use during showers.
For the urine bag, the drainage tube can be customized to your requirements, with adjustable length and width, ensuring leak-proof drainage during emptying and saving you a lot of trouble.
In addition to ostomy bags, we have also developed many accessories, including matching isolation rings, ostomy powder, adhesive release agents, and travel kits. This complete product line provides a comprehensive care solution, all branded with your name, making it convenient for patients and enhancing the trustworthiness of your brand.
Packaging, Brand Promotion, and Compliance
Brand image begins to be conveyed from the first glance at the packaging. We understand that privacy is paramount for ostomy patients. We can customize discreet, inconspicuous outer packaging while clearly indicating the sterility of the contents, ensuring safety while protecting privacy.
Legal compliance is the foundation for entering the market. We ensure that all packaging meets the stringent labeling requirements of major markets worldwide, including FDA regulations in the United States and CE/MDR standards in Europe, among others.

Manufacturing Capability
In the ostomy bag industry, trust isn’t just lip service; it’s a necessity built on solid infrastructure, strict compliance, and precise technology.
Partnering with us means these core strengths will be the strongest foundation for your brand reputation, ensuring that every ostomy bag delivered to patients is safe, effective, and manufactured according to international standards.
Regulatory Compliance and Quality Assurance
In the medical device industry, compliance is essential. It is the cornerstone of private label ostomy bags, a testament to our unwavering commitment to regulatory requirements.Zodelo has established and operates a comprehensive quality management system.
Our production equipment undergoes regular audits, and all necessary documentation for FDA registration in the US and CE marking in Europe is regularly updated.
From raw material warehousing to sterility testing of the final product, every batch undergoes rigorous quality control testing at every stage. We use strict controls throughout the entire process to ensure that every product delivered to patients is stable, safe, and reliable.
Advanced Production Facilities
Our production capacity is determined by state-of-the-art automation technology and precision engineering.
Our factory is equipped with high-precision die-cutting equipment, ensuring that the molding of each adhesive and skin barrier is flawless. This precision is crucial because it is the foundation for preventing leakage and ensuring a perfect fit between the base and the patient’s skin.
To reduce the risk of contamination, we have introduced fully automated assembly lines. This not only significantly reduces human intervention but also ensures that every product strictly complies with regulations.
Research and Development & Materials Science Expertise
Whatever product customization you require, Zodelo‘s technical team can meet your needs, turning patient requirements into tangible, mass-producible, high-quality products.
Scalability and Supply Chain Reliability In business, the worst thing is failing at critical moments and being unable to supply products. Our factory is built for mass production, manufacturing millions of products every month. Whether you’re an industry giant or a startup, we can reliably meet your needs.
We’ve also secured reliable raw material suppliers, ensuring that even amidst market fluctuations, our production lines remain operational and orders are never delayed.
MOQ, Price & Lead Time

Entering the medical device market requires a clear understanding of production and the supply chain.
For example, minimum order quantity (MOQ), pricing, and lead time are all factors that need to be considered from the outset.
We have made reasonable plans for these key aspects.
Minimum Order Quantity (MOQ)
We have categorized MOQs into different tiers, ensuring that both startups and large distributors can find a suitable solution. If you simply want to use our existing products and rebrand them, this is more convenient and faster. You can start with 5,000 to 10,000 pieces per product. If you have unique ideas, such as changing the shape of the chassis, altering the feel of the membrane, or adding a special filter, we need to custom-configure your production line. In this case, the MOQ will be higher, approximately 20,000 to 50,000 pieces, to offset the initial setup costs.
Custom-printed packaging boxes are generally most cost-effective starting with 10,000 pieces. We understand that initial volumes are small, and we can initially meet small-batch production needs through digital printing and other methods.
Pricing Structure
Pricing in the private label ostomy bags is not static, but determined by a precise formula and flexibly adjusted based on specifications and quantities. We employ a transparent tiered pricing model to support your business growth.
Pricing depends first on the product series. Is it a urinary tract ostomy bag or a colostomy bag? Is it a one-piece or two-piece design? Do you need an activated carbon filter or a more comfortable convex base? Different functions and combinations will result in different prices.
The larger the quantity, the greater the discount, and the lower the unit price per product. We have several tiers, such as 10,000 units, 50,000 units, and 100,000 units, with each tier offering a more favorable unit price. This lowers your costs and increases your profits.
Packaging is calculated separately. Using our ready-made packaging is cost-effective; if you want higher-end packaging, the cost will be higher. We will calculate this clearly and include it in your quotation.
Delivery Date
In a highly competitive market, the speed of product launch and distribution is crucial.
For standard private label ostomy bags using existing molds and components, the standard delivery cycle is 30 to 45 days from receiving the final deposit and design confirmation.
If customization requires the development of new molds or material formulations, the delivery time will naturally extend to 60 to 90 days.
The production of customized packaging materials normally proceeds concurrently with product production, and the progress of the printing process will affect the overall schedule. After product production is completed, the final shipping time depends on the logistics method you choose.
How to Start Your Private Label Ostomy Brand

As a professional manufacturer specializing in ostomy bags, we are well aware of the confusion and challenges that newly established private label ostomy bags manufacturers face when entering this market. From product development to compliance implementation, every step is crucial, and mistakes cannot be tolerated.
Based on years of industry experience, we have compiled this comprehensive guide to private-label ostomy bags, from concept to implementation.
I. Define brand positioning: Find your market entry point
Before starting product development, you must first identify your direction. This is like building a house—first lay the foundation; a stable foundation ensures smooth progress.
Figure out who you’re selling to
Ostomy patients have diverse needs; the requirements for urinary ostomy bags differ from those for colostomy bags. Furthermore, you need to first identify your target market. Regulations and competitors vary from place to place, making market research extremely important.
Unique selling points of your product
With so many products on the market, why choose our product? Is it because of better materials, a more discreet design, or higher cost-effectiveness? How can we help people buy a reliable product for less money? Once you’ve identified this selling point, coupled with thoughtful care guidelines and other services, people will remember you immediately.
Define Product Type
Once the direction is set, the product itself is crucial. Should you sell an all-in-one or split-type product? Is a leak-proof design mandatory, or is activated carbon filtration necessary? What’s your target price? Determining these core questions provides a clear roadmap for product development, preventing detours.
II. Select Reliable Manufacturers: Cooperation is the Foundation of Success
Choosing the right manufacturer is a key step to the success of private label ostomy bags.
When evaluating partners, focus on three core dimensions:
Compliance: This is an insurmountable bottom line. Manufacturers must hold ISO 13485 quality management system certification and the FDA registration or CE marking required by the target market to ensure product legality and market access.
Production Capacity: Confirm whether their minimum order quantity (MOQ) matches your budget. Typically, the MOQ for standard products is 5,000 to 10,000 pieces, while highly customized products range from 20,000 to 50,000 pieces. Simultaneously, it’s crucial to define the delivery cycles for standard orders (30-45 days) and custom development (60-90 days) to plan your time-to-market.
Technical Capabilities: Assess whether the manufacturer possesses a professional team capable of supporting material customization, new mold development, and providing clinical application support. This will determine the extent to which your product innovation can be realized.
III. Product Development and Customization: Build Differentiation Competitiveness
Quick Start Mode: This mode prioritizes efficiency. By utilizing existing, mature molds and formulas from manufacturers, you can focus solely on customizing your brand packaging. Its advantages include lower minimum order quantities and shorter time-to-market, making it ideal for brands looking to quickly validate their market presence.
Deep Customization Mode: This path focuses on creating product uniqueness. You can make in-depth adjustments to materials (e.g., adjusting film thickness), shapes (e.g., designing special patch contours), or functions (e.g., integrating smart monitoring modules). While this requires higher minimum order quantities and longer development cycles, it creates a strong competitive advantage.
Product Prototyping and Testing: This is the first step in transforming a concept into a physical product. Rigorous laboratory testing and clinical simulations are required to validate the design’s feasibility.
Technical Parameter Confirmation: Based on prototype testing, the core technical specifications of the product are finalized, such as sealing and skin compatibility, ensuring that product performance meets expectations.
Packaging Design: This is not only a visual representation of the brand but also a crucial compliance step. Packaging design must integrate all regulatory-required labeling information while seamlessly blending into your brand.
IV. Compliance and Quality: A Lifeline That Cannot Be Compromised
Compliance and Quality: Building an Insurmountable Lifeline for Your Brand
In the medical device field, compliance and quality are the crucial foundations for private label ostomy bags, allowing no room for complacency. This requires systematic development across two dimensions: regulatory certification and quality control systems.
Compliance is the only way to enter the market. You must develop a clear certification plan tailored to your target market, such as FDA 510(k) in the US or CE MDR certification in Europe, allowing ample time of 6 to 12 months. The core of this process is preparing a complete and rigorous set of technical documentation, including Design History Files (DHF), Device Master Records (DMR), and Clinical Evaluation Reports (CERs), to demonstrate the product’s safety and effectiveness.
Building a Rigorous Quality Control System to Ensure End-to-End Traceability
Good quality stems from precise control over the entire production process. First, it is essential to ensure that the manufacturing plant strictly adheres to the ISO 13485 quality management system standard, achieving end-to-end traceability from raw material inspection upon arrival, the production process, to final product sterilization. Furthermore, clear checkpoints should be established in key processes, such as airtightness testing and adhesive strength testing, which are crucial for ostomy products, to ensure that every product leaving the factory meets the preset technical parameters.
V.Supply chain and cost management: Balance efficiency and risk
An efficient supply chain and meticulous cost management are key to a brand’s profitability. This requires comprehensive planning at both the production and logistics optimization and pricing strategy levels.
Optimizing Production and Logistics to Build a Resilient Supply Chain
Supply chain resilience is essential for coping with market uncertainties. On the production side, production schedules should be planned in advance with sufficient buffer time to handle unexpected situations such as raw material fluctuations or capacity shortages. On the logistics side, logistics solutions should be flexibly selected based on market urgency and cost budgets—for example, utilizing sea freight to reduce unit costs or using air freight to handle emergency replenishment needs, achieving the optimal balance between cost and efficiency.
Implementing a Tiered Pricing Strategy and Accurately Calculating Total Costs
A clear cost structure is the foundation for pricing decisions. Initial pricing should be based on sales forecasts and negotiated with the manufacturer using tiered pricing, such as locking in tiered unit prices for different orders of 10,000, 50,000, and 100,000 units, leveraging scale for profit. Secondly, a comprehensive total cost accounting model must be established to take into account product costs, regulatory certification fees, logistics costs, and marketing expenses, ensuring that the final price can cover all investments and achieve the expected profit.

FAQs about Private Label Ostomy Bags
Q1. In the ostomy products industry, what is the difference between OEM (Original Equipment Manufacturer) and private label?
OEM focuses on manufacturing to your specifications, while private label emphasizes selling under your brand.
Q2 Which companies are suitable for producing their own-brand ostomy care bags?
Primarily suitable for brand owners, distributors, importers, and cross-border e-commerce sellers of medical products, especially those looking to expand their product lines or increase profit margins.
Q3 Can I use my own brand and packaging?
Yes. Ostomy bags with private labeling support customized logo printing, packaging design, and instruction manuals to meet the brand promotion needs of different markets.
Q4.Does it have the necessary certifications for entering the international market?
ISO 13485, CE, and other certification documents can be provided, depending on the product type and regulatory requirements of the target market.
Q5 What is the Minimum Order Quantity (MOQ)?
Depending on the product type and level of customization, the quantity is typically between 5,000 and 10,000 pieces. The MOQ for standard products is relatively lower.
Q6 Can I get a sample for testing first?
Yes. Samples are used to verify product performance and compatibility, and are an important step before placing a formal order.
Q7: What is the production cycle?
The standard production cycle is 20 to 35 days, depending on order size and the complexity of customization.
Q8. Does this product support customization?
Yes, Zodelo supports personalized customization, including packaging materials, gel systems, and structural designs (such as convex/flat surfaces) to meet the needs of different users.
Q9. How do you ensure consistent product quality?
We ensure product stability and safety through the ISO 13485 quality management system, cleanroom production, and comprehensive quality control.
Q10. What are the core advantages of private-label ostomy bags?
Their advantages lie in lowering the entry barrier, accelerating time-to-market, and helping companies build their own brand, thereby achieving long-term market competitiveness.
9.Conclusion — Private Label Ostomy Bags

At Zodelo,we believe private label ostomy bags should not be cold, impersonal products, but rather a warm expression of care. Our mission is to safeguard patients’ dignity through innovative technology, bringing care back to the warmth of daily life. We listen to our patients, making their needs for comfort and skin-friendliness our primary design principle. We rely on global certifications such as FDA and CE to ensure every aspect of our products is backed by evidence. We collaborate with leading manufacturing partners, starting with 5,000 units to quickly turn good ideas into reality. Leveraging patented technologies such as nanobiomaterials and intelligent leak detection, we give our products a “smart armor,” and based on feedback from the patient community, we make them increasingly user-friendly with continued use.
Our vision is to “redefine the ostomy care experience.” Choosing Zodelo means choosing a lifestyle gently protected by technology and profoundly guided by the brand’s mission—bringing freedom back to patients and making nursing the warmest companionship in daily life.
More Resources about Private Label Ostomy Bags
- A Guide to Ostomy Bags and Pouching Systems— Uchicagomedicine.org
- One Piece Vs Two Piece Stoma Bag: What’S The Difference?– zodelo
- The Ultimate Guide Of Colostomy Bag Size– zodelo
4.Tips for Choosing the Right Ostomy Supplies– uoaa
5.Essential Guide to Choosing Colostomy Bag Suppliers: What You Need to Know– ask
6.How to Choose an Ostomy Bag Manufacturer– zodelo
Advantages of Colostomy Bags: Benefits for Patients and Distributors
Advantages of Colost

What Is a Colostomy Bag Used For?
What Is a Colostomy

OEM vs Standard Ostomy Bags: Which Is Better for Distributors
OEM vs Standard Osto
What Hospitals Look for in an Ostomy Bag Supplier
What Hospitals Look






